Axceler8 Rx: Supporting smarter, more predictable clinical trials
Axceler8 Rx is an innovative clinical trial management platform we developed in partnership with Arthur D. Little (ADL), the world’s first management consulting firm. It is designed to help pharmaceutical companies overcome the complexity of running large-scale studies with greater efficiency and oversight.
By combining Arthur D. Little’s clinical expertise and Vega IT’s experience in building secure, data-intensive digital products, Axceler8 Rx supports smarter planning, more efficient recruitment management, data-driven insights, and stronger compliance, helping sponsors run trials with greater accuracy and confidence.
Clinical trials are lengthy, expensive, and highly complex. Many organisations still rely on outdated tools such as Excel and PowerPoint to manage critical processes, including calculations and estimations of timelines, enrolment, and site capacity. This fragmented approach makes it increasingly difficult to monitor and track progress, especially for large-scale, multi-country studies.
One of the biggest bottlenecks is patient recruitment and retention. It is the leading cause of delays, with research showing that up to 85% of trials experience setbacks. Every delay comes at a high price. In large-scale studies, the cost can reach as much as £0.7 million in lost potential revenue per day, and over £21M lost for each month of delay. This makes accurate forecasting and proactive planning not just valuable, but essential.
On top of this, pharma companies operate in a tightly regulated environment, where compliance is non-negotiable. Outdated systems not only increase the risk of mistakes but also add significant operational and governance overheads, especially around data security, auditability, and change control.
Axceler8 Rx was developed to address these challenges end to end: from trial design and site selection, through forecasting and start-up, to ongoing monitoring and reporting.
Axceler8 Rx helps sponsors answer two core questions early: where to run a clinical trial and how enrolment is likely to evolve over time.
At the core of Axceler8 Rx are four modules that simplify and optimise every major stage of a clinical trial. AI Site Selection and Enrolment Forecast are used for clinical trial modelling and planning, while Recruitment Tool and Site Navigator support clinical trial monitoring, providing visibility into enrolment progress and site-level performance.
1. AI Site Selection
When a pharma company prepares to launch a new clinical trial, one of the first and most complex decisions is where the study can realistically succeed. Selecting sites based on intuition or limited feasibility data often leads to overambitious plans, slow start-up, and enrolment delays.
The AI Site Selection module addresses this challenge by analysing historical clinical trial data for the same or similar indications. It identifies comparable past studies, extracts the sites that previously participated, and presents them as a pool of potential candidates for the new trial.
Building on this foundation, the module allows teams to:
- assess site competition by identifying other ongoing or recent trials targeting the same patient population,
- visualise geographic coverage through maps and isochrones, helping teams understand site accessibility and regional reach,
- enrich the analysis with additional population metrics, such as disease prevalence or age-group distribution at country and site level.
Based on these inputs, the module ranks countries and sites into three tiers (Platinum, Gold, and Silver) and helps teams define an optimal site mix and target number of sites aligned with the desired trial timeline. This enables sponsors to design a site strategy that is both realistic and deliverable, reducing early-stage risk and increasing confidence that enrolment targets can be met within planned timeframes.
2. Enrolment Forecast
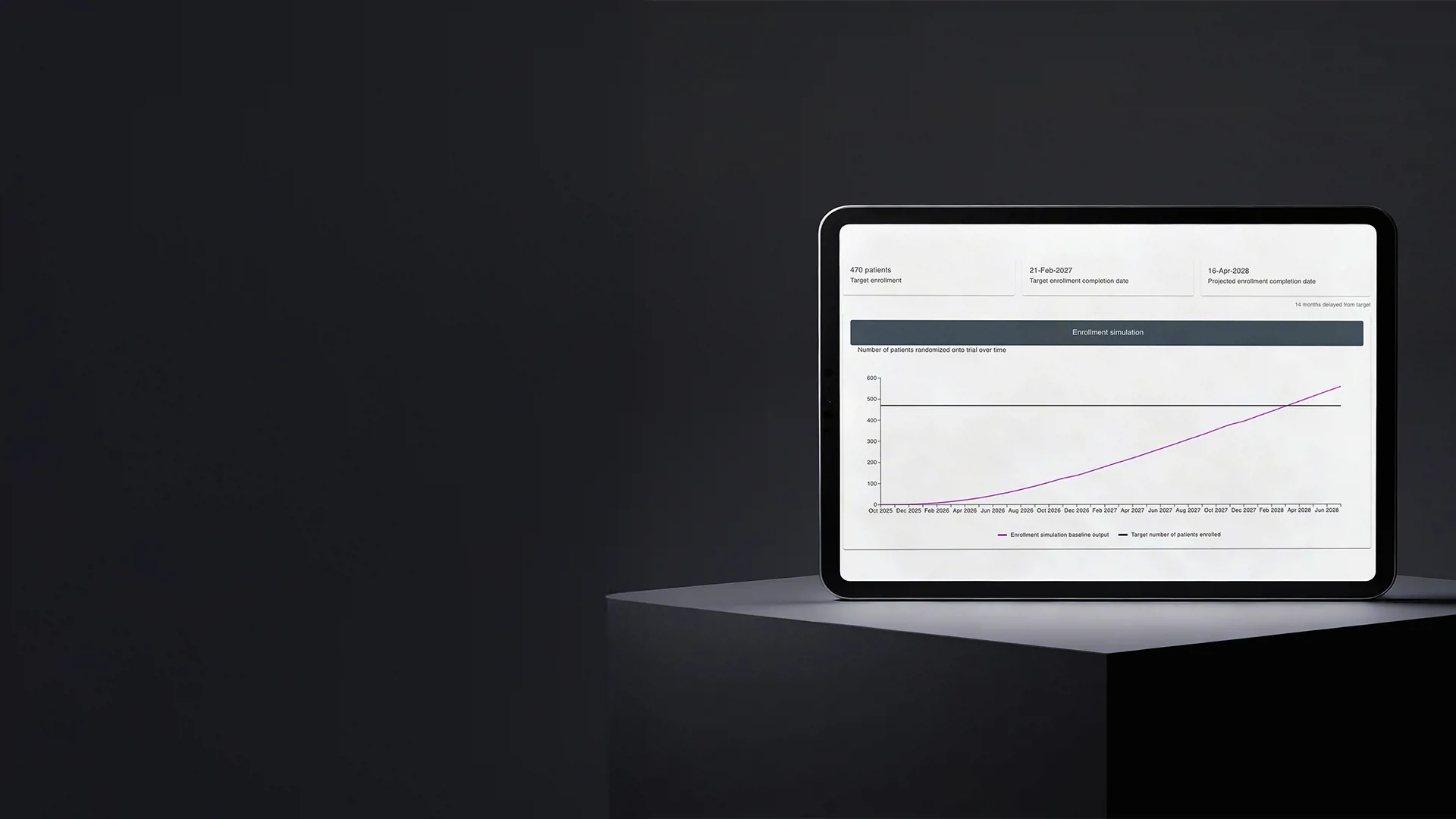
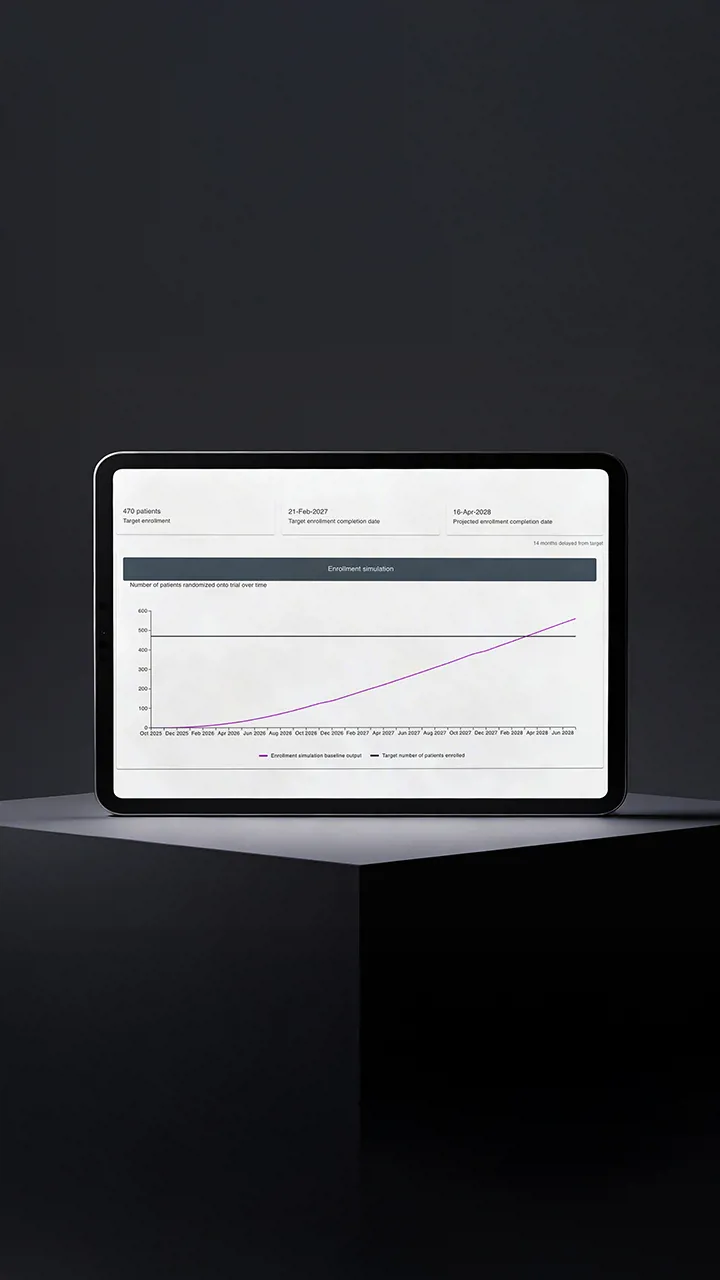
The core problem the Enrolment Forecast module addresses is providing a realistic projection of how a clinical trial is likely to progress and what outcomes can be expected over time.
The module is used in both phases. During trial planning, it works with limited inputs such as indication, early feasibility assumptions, and historical experience from similar studies. Based on this data, it generates projected enrolment curves that help teams test assumptions and understand how different site, country, or criteria configurations may affect timelines.
Once recruitment begins, the module incorporates live trial data, including the number of active sites and enrolled patients. Forecasts are recalculated from the current state of the study, producing increasingly accurate projections as more data becomes available.
At the core of the module is a Monte Carlo simulation, which evaluates thousands of possible enrolment scenarios to determine the most likely trajectory and associated risk. This gives sponsors a realistic view of how the trial is progressing and what outcomes they can expect under current conditions.
3. Site Navigator
The core problem Site Navigator addresses is the complexity and unpredictability of site activation, one of the largest sources of delay in clinical trials.
Before a site can enrol its first patient, it must complete multiple interdependent activities, including regulatory approvals, contracts, staff training, vendor coordination, and the availability of specialised equipment.
The Site Navigator module provides study teams with a clear, structured view of every step required to activate a site, from initial readiness through ongoing performance. Each activity is tracked as part of a broader activation flow, making it easy to see where progress is slowing down.
It helps teams quickly identify where delays are occurring, whether at country level, site level, or within specific tasks. For example, it can highlight missing documentation, incomplete training, or dependencies on external vendors, such as the delivery of specialised equipment required to begin patient treatment.
By breaking activation into traceable steps, Site Navigator enables timely intervention and better coordination across teams, helping sites reach first-patient-in in a more controlled and auditable way.
4. Trial Dashboard Suite
The Trial Dashboard Suite addresses a common challenge in clinical trials: having a clear, unified view of multiple studies and their progress without relying on fragmented reports and manual cross-referencing.
It supports ongoing trial monitoring by tracking enrolment progress, data collection status, and compliance milestones across sites and countries. Study teams can compare actual enrolment against forecasts, identify deviations early, and adjust recruitment strategies as trials progress.
The suite brings together a set of focused dashboards, each designed to surface insights for different audiences and operational needs. By consolidating information at program, study, site, country, and participant level, it provides a consistent overview of trial performance while still allowing teams to drill into specific areas when needed.
Key operational metrics and KPIs are calculated and presented in a single place, eliminating the need to manually combine data from multiple sources. This allows stakeholders to quickly understand how trials are progressing, where delays are emerging, and which areas require attention, supporting more informed and timely decision-making.
Data Speak: AI-powered trial intelligence
Data Speak is Axceler8 Rx’s AI assistant for clinical trials.
It turns data into instant answers by allowing users to ask questions in natural language and get insights across all dashboards and modules, from site performance to patient trends.
Typical use cases include:
- “Which countries are behind the target for this study and why?”
- “Show me sites with the highest screen failure rates in the last 30 days.”
- “Which sites are most similar to our top performers in terms of indication and geography?”
Data Speak positions Axceler8 Rx as a central hub for trial intelligence, reducing the need for manual reporting and allowing study teams to get to decisions faster.
Data integration, security and compliance
We developed Axceler8 Rx as a single-tenant platform that runs inside each client’s environment, because every pharma organisation works with its own systems, data formats, and governance rules.This makes integration a critical first step, creating a single, trusted source of trial data and enabling accurate modelling, forecasting, and reporting.
To support this, we built adaptable data templates and integration flows that map to any client data source, whether spreadsheets, internal applications, document repositories, or synchronised data zones in environments where outbound API calls are restricted.
For maximum security and compliance, Axceler8 Rx never exposes production data in development, follows strict change management and validation processes, and includes built-in fallback and rollback mechanisms. This architecture allows the platform to meet the high data protection and quality standards required by global pharma organisations.
We implemented Axceler8 Rx with our first enterprise pharmaceutical client as a single-tenant application deployed within the client’s environment. The rollout was delivered in two phases, allowing us to introduce the platform gradually and refine it based on real-world usage.
As part of the implementation, we delivered and integrated three core modules: AI Site Selection, Enrolment Forecast, and Site Navigator. These modules enabled the client to model, forecast, and initiate clinical studies directly within Axceler8 Rx, while gaining clearer visibility into site performance and enrolment dynamics.
As adoption progressed, we continued to evolve the platform alongside the client. We introduced additional integrations and extended existing modules to better reflect real clinical trial requirements. One of the key enhancements during this period was the introduction of patient categorisation, allowing studies to distinguish between different patient groups (for example, by disease stage or treatment line). This required updates across the data model and reporting, enabling more accurate and granular analysis.
Insights gained throughout both phases were incorporated back into the core product. This strengthened Axceler8 Rx and helped us establish a more scalable and repeatable rollout model for future enterprise clients.
Key lessons
- Enterprise pharma environments impose strict integration constraints
Through real-world deployment, we confirmed the importance of designing clinical trial platforms that operate fully within controlled client environments and comply with established governance and data-access rules.
- Core trial workflows require robust validation
Supporting processes such as trial initiation and forecasting reinforced the need for strong validation at every workflow step to ensure reliability in regulated settings.
- Real-world usage drives product maturity
Feedback from deploying Axceler8 Rx in practice informed product improvements that strengthened its foundations and readiness for future implementations.
Axceler8 Rx strengthens how pharma companies plan, monitor, and execute clinical trials. By bringing modelling, forecasting, site management, and AI-driven insights into a unified platform, sponsors and CROs gain clearer oversight, faster decision-making, and more predictable study outcomes.
The platform delivers:
- More accurate planning and forecasting through scenario modelling, site selection, and Monte Carlo projections that help teams design realistic and achievable study plans.
- Better operational oversight, with real-time tracking that keeps trials closer to schedule and budget.
- Fewer delays and lower costs, thanks to earlier detection of bottlenecks and streamlined workflows across teams and sites.
- A scalable, secure foundation, with single-tenant deployment that ensures data isolation, integrity, and compliance with strict pharma governance.
- Faster access to insights, as Data Speak removes the need for manual reporting and reduces analytical workload.
- Stronger compliance, supported by controlled integrations and strict separation of production data.
The outcome: more predictable trials, reduced operational effort, and faster delivery of life-changing treatments to patients.
Built on 15+ years of experience across 100+ studies, Axceler8 Rx modules have already been used in 30+ clinical trials. The results show a clear impact:
- 81% accuracy in site activation projections across 50+ countries
- 95% accuracy in bottom-up enrolment forecasts vs. actuals
- 300+ additional qualified sites identified by the AI Site Selection module compared to standard feasibility methods.
These outcomes lead to more predictable timelines, better utilisation of high-performing sites, reduced risk of under-recruitment and ultimately, faster and more efficient delivery of treatments to patients.
Site Selection
Data Integration:
- Python
- Spark
- SQL
- PostgreSQL
- AWS Glue
- AWS S3
- AWS Step Functions
- AWS Lambda
Enrollment Forecasting & Site Navigator
Backend:
- Python 3.12+
- Flask 3.x
- Flask-SQLAlchemy / SQLAlchemy 2.0
- PostgreSQL
- Pydantic 2.x
- Gunicorn
Frontend:
- React 19
- Typescript
- Vite 7
- MUI
- Plotly
- MUI X Charts
Data Science:
- Pandas 2.x
- NumPy 2.x
- SciPy
Development Tools:
- Ruff
- Mypy
- Pytest
- Playwright